-
Actiq (Cephalon)
WARNING: May be habit formingPHYSICIANS AND OTHER HEALTHCARE PROVIDERS MUST BECOME FAMILIAR WITH THE IMPORTANT WARNINGS IN THIS LABEL .
Actiq is indicated only for the management of breakthrough cancer pain in patients with malignancies who are already receiving and who are tolerant to opioid therapy for their underlying persistent cancer pain.
Patients considered opioid tolerant are those who are taking at least 60 mg morphine/day, 50 mcg transdermal fentanyl/hour, or an equianalgesic dose of another opioid for a week or longer.
Because life-threatening hypoventilation could occur at any dose in patients not taking chronic opiates, Actiq is contraindicated in the management of acute or postoperative pain. This product must not be used in opioid non-tolerant patients.
Actiq is intended to be used only in the care of cancer patients and only by oncologists and pain specialists who are knowledgeable of and skilled in the use of Schedule II opioids to treat cancer pain.
Patients and their caregivers must be instructed that Actiq contains a medicine in an amount which can be fatal to a child. Patients and their caregivers must be instructed to keep all units out of the reach of children and to discard opened units properly. (See Information for Patients and Their Caregivers for disposal instructions.)
DESCRIPTION
Actiq (oral transmucosal fentanyl citrate) is a solid formulation of fentanyl citrate, a potent opioid analgesic, intended for oral transmucosal administration. Actiq is formulated as a white to off-white solid drug matrix on a handle that is radiopaque and is fracture resistant (ABS plastic) under normal conditions when used as directed.
Actiq is designed to be dissolved slowly in the mouth in a manner to facilitate transmucosal absorption. The handle allows the Actiq unit to be removed from the mouth if signs of excessive opioid effects appear during administration.
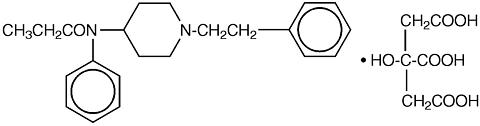
Active Ingredient: Fentanyl citrate, USP is N-(1-Phenethyl-4-piperidyl) propionanilide citrate (1:1). Fentanyl is a highly lipophilic compound (octanol-water partition coefficient at pH 7.4 is 816:1) that is freely soluble in organic solvents and sparingly soluble in water (1:40). The molecular weight of the free base is 336.5 (the citrate salt is 528.6). The pKa of the tertiary nitrogens are 7.3 and 8.4. The compound has the following structural formula:
Actiq is available in six strengths equivalent to 200, 400, 600, 800, 1200, or 1600 mcg fentanyl base that is identified by the text on the solid drug matrix, the dosage unit handle tag, the blister package, and the shelf carton.
Inactive Ingredients: Hydrated dextrates, citric acid, dibasic sodium phosphate, artificial berry flavor, magnesium stearate, modified food starch, and confectioner's sugar.
CLINICAL PHARMACOLOGY AND PHARMACOKINETICS
Pharmacology:
Fentanyl, a pure opioid agonist, acts primarily through interaction with opioid mu-receptors located in the brain, spinal cord and smooth muscle. The primary site of therapeutic action is the central nervous system (CNS). The most clinically useful pharmacologic effects of the interaction of fentanyl with mu-receptors are analgesia and sedation.
Other opioid effects may include somnolence, hypoventilation, bradycardia, postural hypotension, pruritus, dizziness, nausea, diaphoresis, flushing, euphoria and confusion or difficulty in concentrating at clinically relevant doses.
Clinical Pharmacology
Analgesia:
The analgesic effects of fentanyl are related to the blood level of the drug, if proper allowance is made for the delay into and out of the CNS (a process with a 3-to-5-minute half-life). In opioid non-tolerant individuals, fentanyl provides effects ranging from analgesia at blood levels of 1 to 2 ng/mL, all the way to surgical anesthesia and profound respiratory depression at levels of 10-20 ng/mL.
In general, the minimum effective concentration and the concentration at which toxicity occurs rise with increasing tolerance to any and all opioids. The rate of development of tolerance varies widely among individuals. As a result, the dose of Actiq should be individually titrated to achieve the desired effect (see DOSAGE AND ADMINISTRATION ).
Gastrointestinal (GI) Tract and Other Smooth Muscle:
Opioids increase the tone and decrease contractions of the smooth muscle of the gastrointestinal (GI) tract. This results in prolongation in GI transit time and may be responsible for the constipating effect of opioids. Because opioids may increase biliary tract pressure, some patients with biliary colic may experience worsening of pain.
While opioids generally increase the tone of urinary tract smooth muscle, the overall effect tends to vary, in some cases producing urinary urgency, in others, difficulty in urination.
Respiratory System:
All opioid mu-receptor agonists, including fentanyl, produce dose dependent respiratory depression. The risk of respiratory depression is less in patients receiving chronic opioid therapy who develop tolerance to respiratory depression and other opioid effects. During the titration phase of the clinical trials, somnolence, which may be a precursor to respiratory depression, did increase in patients who were treated with higher doses of Actiq. In studies of opioid non-tolerant subjects, respiratory rate and oxygen saturation typically decrease as fentanyl blood concentration increases. Typically, peak respiratory depressive effects (decrease in respiratory rate) are seen 15 to 30 minutes from the start of oral transmucosal fentanyl citrate (OTFC®) administration and may persist for several hours.
Serious or fatal respiratory depression can occur, even at recommended doses, in vulnerable individuals. As with other potent opioids, fentanyl has been associated with cases of serious and fatal respiratory depression in opioid non-tolerant individuals.
Fentanyl depresses the cough reflex as a result of its CNS activity. Although not observed with Actiq in clinical trials, fentanyl given rapidly by intravenous injection in large doses may interfere with respiration by causing rigidity in the muscles of respiration. Therefore, physicians and other healthcare providers should be aware of this potential complication. (See BOX WARNING, CONTRAINDICATIONS, WARNINGS, PRECAUTIONS, ADVERSE REACTIONS, and OVERDOSAGE for additional information on hypoventilation.)
Pharmacokinetics
Absorption:
The absorption pharmacokinetics of fentanyl from the oral transmucosal dosage form is a combination of an initial rapid absorption from the buccal mucosa and a more prolonged absorption of swallowed fentanyl from the GI tract. Both the blood fentanyl profile and the bioavailability of fentanyl will vary depending on the fraction of the dose that is absorbed through the oral mucosa and the fraction swallowed.
Absolute bioavailability, as determined by area under the concentration-time curve, of 15 mcg/kg in 12 adult males was 50% compared to intravenous fentanyl.
Normally, approximately 25% of the total dose of Actiq is rapidly absorbed from the buccal mucosa and becomes systemically available. The remaining 75% of the total dose is swallowed with the saliva and then is slowly absorbed from the GI tract. About 1/3 of this amount (25% of the total dose) escapes hepatic and intestinal first-pass elimination and becomes systemically available. Thus, the generally observed 50% bioavailability of Actiq is divided equally between rapid transmucosal and slower GI absorption. Therefore, a unit dose of Actiq, if chewed and swallowed, might result in lower peak concentrations and lower bioavailability than when consumed as directed.
Dose proportionality among four of the available strengths of Actiq (200, 400, 800, and 1600 mcg) has been demonstrated in a balanced crossover design in adult subjects. Mean serum fentanyl levels following these four doses of Actiq are shown in Figure 1. The curves for each dose level are similar in shape with increasing dose levels producing increasing serum fentanyl levels. C max and AUC 0 ->(infinity) increased in a dose-dependent manner that is approximately proportional to the Actiq administered.
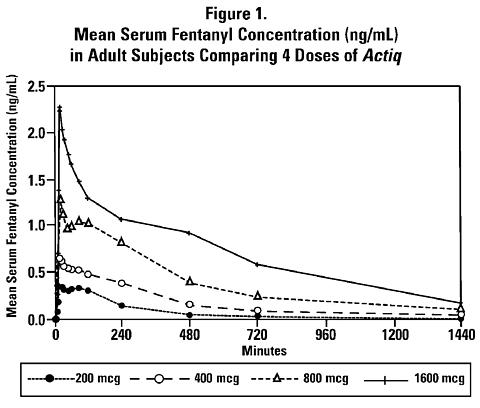
The pharmacokinetic parameters of the four strengths of Actiq tested in the dose-proportionality study are shown in Table 1. The mean C max ranged from 0.39-2.51 ng/mL. The median time of maximum plasma concentration (T max ) across these four doses of Actiq varied from 20-40 minutes (range of 20-480 minutes) as measured after the start of administration.
Table 1.
Pharmacokinetic Parameters in Adult Subjects Receiving
200, 400, 800, and 1600 mcg Units of ActiqPharmacokinetic Parameter 200 mcg 400 mcg 800 mcg 1600 mcg T max , minute median (range) 40 (20-120) 25 (20-240) 25 (20-120) 20 (20-480) C max , ng/mL mean (% CV) 0.39 (23) 0.75 (33) 1.55 (30) 2.51 (23) AUC 0-1440 , ng/mL minute mean (% CV) 102 (65) 243 (67) 573 (64) 1026 (67) t ½ , minute mean (% CV) 193 (48) 386 (115) 381 (55) 358 (45)
Distribution:
Fentanyl is highly lipophilic. Animal data showed that following absorption, fentanyl is rapidly distributed to the brain, heart, lungs, kidneys and spleen followed by a slower redistribution to muscles and fat. The plasma protein binding of fentanyl is 80-85%. The main binding protein is alpha-1-acid glycoprotein, but both albumin and lipoproteins contribute to some extent. The free fraction of fentanyl increases with acidosis. The mean volume of distribution at steady state (Vss) was 4 L/kg.
Metabolism:
Fentanyl is metabolized in the liver and in the intestinal mucosa to norfentanyl by cytochrome P450 3A4 isoform. Norfentanyl was not found to be pharmacologically active in animal studies (see PRECAUTIONS: Drug Interactions for additional information).
Elimination:
Fentanyl is primarily (more than 90%) eliminated by biotransformation to N-dealkylated and hydroxylated inactive metabolites. Less than 7% of the dose is excreted unchanged in the urine, and only about 1% is excreted unchanged in the feces. The metabolites are mainly excreted in the urine, while fecal excretion is less important. The total plasma clearance of fentanyl was 0.5 L/hr/kg (range 0.3-0.7 L/hr/kg). The terminal elimination half-life after OTFC administration is about 7 hours.
Special Populations:
Elderly Patients:
Elderly patients have been shown to be twice as sensitive to the effects of fentanyl when administered intravenously, compared with the younger population. While a formal study evaluating the safety profile of Actiq in the elderly population has not been performed, in the 257 opioid tolerant cancer patients studied with Actiq , approximately 20% were over age 65 years. No difference was noted in the safety profile in this group compared to those aged less than 65 years, though they did titrate to lower doses than younger patients (see PRECAUTIONS ).
Patients with Renal or Hepatic Impairment:
Actiq should be administered with caution to patients with liver or kidney dysfunction because of the importance of these organs in the metabolism and excretion of drugs and effects on plasma-binding proteins (see PRECAUTIONS ).
Although fentanyl kinetics are known to be altered in both hepatic and renal disease due to alterations in metabolic clearance and plasma proteins, individualized doses of Actiq have been used successfully for breakthrough cancer pain in patients with hepatic and renal disorders. The duration of effect for the initial dose of fentanyl is determined by redistribution of the drug, such that diminished metabolic clearance may only become significant with repeated dosing or with excessively large single doses. For these reasons, while doses titrated to clinical effect are recommended for all patients, special care should be taken in patients with severe hepatic or renal disease.
Gender
Both male and female opioid-tolerant cancer patients were studied for the treatment of breakthrough cancer pain. No clinically relevant gender differences were noted either in dosage requirement or in observed adverse events.
CLINICAL TRIALS
Breakthrough Cancer Pain:
Actiq was investigated in clinical trials involving 257 opioid tolerant adult cancer patients experiencing breakthrough cancer pain. Breakthrough cancer pain was defined as a transient flare of moderate-to-severe pain occurring in cancer patients experiencing persistent cancer pain otherwise controlled with maintenance doses of opioid medications including at least 60 mg morphine/day, 50 mcg transdermal fentanyl/hour, or an equianalgesic dose of another opioid for a week or longer.
In two dose titration studies 95 of 127 patients (75%) who were on stable doses of either long-acting oral opioids or transdermal fentanyl for their persistent cancer pain titrated to a successful dose of Actiq to treat their breakthrough cancer pain within the dose range offered (200, 400, 600, 800, 1200 and 1600 mcg). In these studies 11% of patients withdrew due to adverse events and 14% withdrew due to other reasons. A "successful" dose was defined as a dose where one unit of Actiq could be used consistently for at least two consecutive days to treat breakthrough cancer pain without unacceptable side effects.
The successful dose of Actiq for breakthrough cancer pain was not predicted from the daily maintenance dose of opioid used to manage the persistent cancer pain and is thus best determined by dose titration.
A double-blind placebo controlled crossover study was performed in cancer patients to evaluate the effectiveness of Actiq for the treatment of breakthrough cancer pain. Of 130 patients who entered the study 92 patients (71%) achieved a successful dose during the titration phase. The distribution of successful doses is shown in Table 2.
Table 2.
Successful Dose of Actiq
Following Initial TitrationTotal No (%) Actiq Dose (N=92) 200 mcg 13 (14) 400 mcg 19 (21) 600 mcg 14 (15) 800 mcg 18 (20) 1200 mcg 13 (14) 1600 mcg 15 (16) Mean±SD 789±468 mcg
On average, patients over 65 years of age titrated to a mean dose that was about 200 mcg less than the mean dose to which younger adult patients were titrated.
Actiq produced statistically significantly more pain relief compared with placebo at 15, 30, 45 and 60 minutes following administration (see Figure 2).
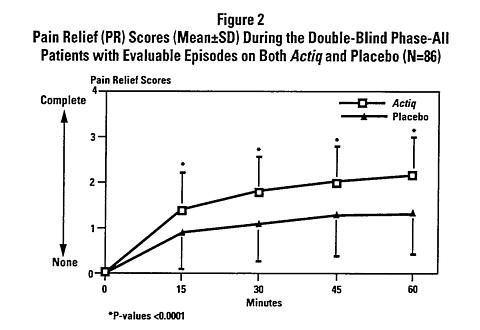
In this same study patients also rated the performance of medication to treat their breakthrough cancer pain using a different scale ranging from "poor" to "excellent." On average, placebo was rated "fair" and Actiq was rated "good."
INDICATIONS AND USAGE
(See BOX WARNING and CONTRAINDICATIONS)
Actiq is indicated only for the management of breakthrough cancer pain in patients with malignancies who are already receiving and who are tolerant to opioid therapy for their underlying persistent cancer pain. Patients considered opioid tolerant are those who are taking at least 60 mg morphine/day, 50 mcg transdermal fentanyl/hour, or an equianalgesic dose of another opioid for a week or longer.
Because life-threatening hypoventilation could occur at any dose in patients not taking chronic opiates, Actiq is contraindicated in the management of acute or postoperative pain. This product must not be used in opioid non-tolerant patients.
Actiq is intended to be used only in the care of cancer patients and only by oncologists and pain specialists who are knowledgeable of and skilled in the use of Schedule II opioids to treat cancer pain.
Actiq should be individually titrated to a dose that provides adequate analgesia and minimizes side effects. If signs of excessive opioid effects appear before the unit is consumed, the dosage unit should be removed from the patient's mouth immediately, disposed of properly, and subsequent doses should be decreased (see DOSAGE AND ADMINISTRATION ).
Patients and their caregivers must be instructed that Actiq contains a medicine in an amount that can be fatal to a child. Patients and their caregivers must be instructed to keep all units out of the reach of children and to discard opened units properly in a secured container.
CONTRAINDICATIONS
Because life-threatening hypoventilation could occur at any dose in patients not taking chronic opiates, Actiq is contraindicated in the management of acute or postoperative pain. The risk of respiratory depression begins to increase with fentanyl plasma levels of 2.0 ng/mL in opioid non-tolerant individuals (see Pharmacokinetics). This product must not be used in opioid non-tolerant patients.
Patients considered opioid tolerant are those who are taking at least 60 mg morphine/day, 50 mcg transdermal fentanyl/hour, or an equianalgesic dose of another opioid for a week or longer.
Actiq is contraindicated in patients with known intolerance or hypersensitivity to any of its components or the drug fentanyl.
WARNINGS
See BOX WARNING .)
The concomitant use of other CNS depressants, including other opioids, sedatives or hypnotics, general anesthetics, phenothiazines, tranquilizers, skeletal muscle relaxants, sedating antihistamines, potent inhibitors of cytochrome P450 3A4 isoform (e.g., erythromycin, ketoconazole, and certain protease inhibitors), and alcoholic beverages may produce increased depressant effects. Hypoventilation, hypotension, and profound sedation may occur.
Actiq is not recommended for use in patients who have received MAO inhibitors within 14 days, because severe and unpredictable potentiation by MAO inhibitors has been reported with opioid analgesics.
Pediatric Use: The appropriate dosing and safety of Actiq in opioid tolerant children with breakthrough cancer pain have not been established below the age of 16 years.
Patients and their caregivers must be instructed that Actiq contains a medicine in an amount which can be fatal to a child. Patients and their caregivers must be instructed to keep both used and unused dosage units out of the reach of children. While all units should be disposed of immediately after use, partially consumed units represent a special risk to children. In the event that a unit is not completely consumed it must be properly disposed as soon as possible. (See SAFETY AND HANDLING, PRECAUTIONS, and PATIENT LEAFLET for specific patient instructions.)
Physicians and dispensing pharmacists must specifically question patients or caregivers about the presence of children in the home on a full time or visiting basis and counsel them regarding the dangers to children from inadvertent exposure.
PRECAUTIONS
General
The initial dose of Actiq to treat episodes of breakthrough cancer pain should be 200 mcg. Each patient should be individually titrated to provide adequate analgesia while minimizing side effects.
Opioid analgesics impair the mental and/or physical ability required for the performance of potentially dangerous tasks (e.g., driving a car or operating machinery). Patients taking Actiq should be warned of these dangers and should be counseled accordingly.
The use of concomitant CNS active drugs requires special patient care and observation. (See WARNINGS .)
Hypoventilation (Respiratory Depression)
As with all opioids, there is a risk of clinically significant hypoventilation in patients using Actiq . Accordingly, all patients should be followed for symptoms of respiratory depression. Hypoventilation may occur more readily when opioids are given in conjunction with other agents that depress respiration.
Chronic Pulmonary Disease
Because potent opioids can cause hypoventilation, Actiq should be titrated with caution in patients with chronic obstructive pulmonary disease or pre-existing medical conditions predisposing them to hypoventilation. In such patients, even normal therapeutic doses of Actiq may further decrease respiratory drive to the point of respiratory failure.
HEAD INJURIES AND INCREASED INTRACRANIAL PRESSURE
Actiq should only be administered with extreme caution in patients who may be particularly susceptible to the intracranial effects of CO 2 retention such as those with evidence of increased intracranial pressure or impaired consciousness. Opioids may obscure the clinical course of a patient with a head injury and should be used only if clinically warranted.
Cardiac Disease
Intravenous fentanyl may produce bradycardia. Therefore, Actiq should be used with caution in patients with bradyarrhythmias.
Hepatic or Renal Disease
Actiq should be administered with caution to patients with liver or kidney dysfunction because of the importance of these organs in the metabolism and excretion of drugs and effects on plasma binding proteins (see PHARMACOKINETICS ).
Information for Patients and Their Caregivers
Patients and their caregivers must be instructed that Actiq contains medicine in an amount that could be fatal to a child. Patients and their caregivers must be instructed to keep both used and unused dosage units out of the reach of children. Partially consumed units represent a special risk to children. In the event that a unit is not completely consumed it must be properly disposed as soon as possible. (See SAFETY AND HANDLING, WARNINGS, and PATIENT LEAFLET for specific patient instructions.)
Frequent consumption of sugar-containing products may increase the risk of dental decay (each Actiq unit contains approximately 2 grams of sugar {hydrated dextrates}). The occurrence of dry mouth associated with the use of opioid medications (such as fentanyl) may add to this risk.
Post-marketing reports of dental decay have been received in patients taking Actiq (see ADVERSE REACTIONS -- Post Marketing Experience ). In some of these patients, dental decay occurred despite reported routine oral hygiene. Therefore, patients using Actiq should consult their dentist to ensure appropriate oral hygiene.
Diabetic patients should be advised that Actiq contains approximately 2 grams of sugar per unit.
Patients and their caregivers should be provided with an Actiq Welcome Kit, which contains educational materials and safe storage containers to help patients store Actiq and other medicines out of the reach of children. Patients and their caregivers should also have an opportunity to watch the patient safety video, which provides proper product use, storage, handling and disposal directions. Patients should also have an opportunity to discuss the video with their health care providers. Health care professionals should call 1-800-896-5855 to obtain a supply of welcome kits or videos for patient viewing.
Disposal of Used Actiq Units
Patients must be instructed to dispose of completely used and partially used Actiq units.
- After consumption of the unit is complete and the matrix is totally dissolved, throw away the handle in a trash container that is out of the reach of children.
- If any of the drug matrix remains on the handle, place the handle under hot running tap water until all of the drug matrix is dissolved, and then dispose of the handle in a place that is out of the reach of children.
- Handles in the child-resistant container should be disposed of (as described in steps 1 and 2) at least once a day.
If the patient does not entirely consume the unit and the remaining drug cannot be immediately dissolved under hot running water, the patient or caregiver must temporarily store the Actiq unit in the specially provided child-resistant container out of the reach of children until proper disposal is possible.
Disposal of Unopened Actiq Units When No Longer Needed
Patients and members of their household must be advised to dispose of any unopened units remaining from a prescription as soon as they are no longer needed.
To dispose of the unused Actiq units:
- Remove the Actiq unit from its blister package using scissors, and hold the Actiq by its handle over the toilet bowl.
- Using wire-cutting pliers cut off the drug matrix end so that it falls into the toilet.
- Dispose of the handle in a place that is out of the reach of children.
- Repeat steps 1, 2, and 3 for each Actiq unit. Flush the toilet twice after 5 units have been cut and deposited into the toilet.
Do not flush the entire Actiq units, Actiq handles, blister packages, or cartons down the toilet. The handle should be disposed of where children cannot reach it (see SAFETY AND HANDLING ).
Detailed instructions for the proper storage, administration, disposal, and important instructions for managing an overdose of Actiq are provided in the Actiq Patient Leaflet. Patients should be encouraged to read this information in its entirety and be given an opportunity to have their questions answered.
In the event that a caregiver requires additional assistance in disposing of excess unusable units that remain in the home after a patient has expired, they should be instructed to call the toll-free number (1-800-896-5855) or seek assistance from their local DEA office.
Laboratory Tests
The effects of Actiq on laboratory tests have not been evaluated.
Drug Interactions
See WARNINGS .
Fentanyl is metabolized in the liver and intestinal mucosa to norfentanyl by the cytochrome P450 3A4 isoform. Drugs that inhibit P450 3A4 activity may increase the bioavailability of swallowed fentanyl (by decreasing intestinal and hepatic first pass metabolism) and may decrease the systemic clearance of fentanyl. The expected clinical results would be increased or prolonged opioid effects. Drugs that induce cytochrome P450 3A4 activity may have the opposite effects. However, no in vitro or in vivo studies have been performed to assess the impact of those potential interactions on the administration of Actiq. Thus patients who begin or end therapy with potent inhibitors of CYP450 3A4 such as macrolide antibiotics (e.g., erythromycin), azole antifungal agents (e.g., ketoconazole and itraconazole), and protease inhibitors (e.g., ritanovir) while receiving Actiq should be monitored for a change in opioid effects and, if warranted, the dose of Actiq should be adjusted.
Carcinogenesis, Mutagenesis, and Impairment of Fertility
Because animal carcinogenicity studies have not been conducted with fentanyl citrate, the potential carcinogenic effect of Actiq is unknown.
Standard mutagenicity testing of fentanyl citrate has been conducted. There was no evidence of mutagenicity in the Ames Salmonella or Escherichia mutagenicity assay, the in-vitro mouse lymphoma mutagenesis assay, and the in-vivo micronucleus cytogenetic assay in the mouse.
Reproduction studies in rats revealed a significant decrease in the pregnancy rate of all experimental groups. This decrease was most pronounced in the high dose group (1.25 mg/kg subcutaneously) in which one of twenty animals became pregnant.
Pregnancy--Category C
Fentanyl has been shown to impair fertility and to have an embryocidal effect with an increase in resorptions in rats when given for a period of 12 to 21 days in doses of 30 mcg/kg IV or 160 mcg/kg subcutaneously.
No evidence of teratogenic effects has been observed after administration of fentanyl citrate to rats. There are no adequate and well-controlled studies in pregnant women. Actiq should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Labor and Delivery
Actiq is not indicated for use in labor and delivery.
Nursing Mothers
Fentanyl is excreted in human milk; therefore Actiq should not be used in nursing women because of the possibility of sedation and/or respiratory depression in their infants.
Pediatric Use
See WARNINGS
Geriatric Use
Of the 257 patients in clinical studies of Actiq in breakthrough cancer pain, 61 (24%) were 65 and over, while 15 (6%) were 75 and over.
Those patients over the age of 65 titrated to a mean dose that was about 200 mcg less than the mean dose titrated to by younger patients. Previous studies with intravenous fentanyl showed that elderly patients are twice as sensitive to the effects of fentanyl as the younger population.
No difference was noted in the safety profile of the group over 65 as compared to younger patients in Actiq clinical trials. However, greater sensitivity in older individuals cannot be ruled out. Therefore, caution should be exercised in individually titrating Actiq in elderly patients to provide adequate efficacy while minimizing risk.
ADVERSE REACTIONS
Pre-Marketing Clinical Trial Experience
The safety of Actiq has been evaluated in 257 opioid tolerant chronic cancer pain patients. The duration of Actiq use varied during the open-label study. Some patients were followed for over 21 months. The average duration of therapy in the open-label study was 129 days.
The adverse events seen with Actiq are typical opioid side effects. Frequently, these adverse events will cease or decrease in intensity with continued use of Actiq, as the patient is titrated to the proper dose. Opioid side effects should be expected and managed accordingly.
The most serious adverse effects associated with all opioids are respiratory depression (potentially leading to apnea or respiratory arrest), circulatory depression, hypotension, and shock. All patients should be followed for symptoms of respiratory depression.
Because the clinical trials of Actiq were designed to evaluate safety and efficacy in treating breakthrough cancer pain, all patients were also taking concomitant opioids, such as sustained-release morphine or transdermal fentanyl, for their persistent cancer pain. The adverse event data presented here reflect the actual percentage of patients experiencing each adverse effect among patients who received Actiq for breakthrough cancer pain along with a concomitant opioid for persistent cancer pain. There has been no attempt to correct for concomitant use of other opioids, duration of Actiq therapy, or cancer-related symptoms. Adverse events are included regardless of causality or severity.
Three short-term clinical trials with similar titration schemes were conducted in 257 patients with malignancy and breakthrough cancer pain. Data are available for 254 of these patients. The goal of titration in these trials was to find the dose of Actiq that provided adequate analgesia with acceptable side effects (successful dose). Patients were titrated from a low dose to a successful dose in a manner similar to current titration dosing guidelines. Table 3 lists by dose groups, adverse events with an overall frequency of 1% or greater that occurred during titration and are commonly associated with opioid administration or are of particular clinical interest. The ability to assign a dose-response relationship to these adverse events is limited by the titration schemes used in these studies. Adverse events are listed in descending order of frequency within each body system.
Table 3.
Percent of Patients with Specific Adverse Events Commonly
Associated with Opioid Administration or of Particular Clinical Interest
Which Occurred During Titration (Events in 1% or More of Patients)Dose Group 200-600 mcg 800-1400 mcg 1600 mcg >1600 mcg Any Number of Patients 230 138 54 41 254 Body As A Whole Asthenia 6 4 0 7 9 Headache 3 4 6 5 6 Accidental Injury 1 1 4 0 2 Digestive Nausea 14 15 11 22 23 Vomiting 7 6 6 15 12 Constipation 1 4 2 0 4 Nervous Dizziness 10 16 6 15 17 Somnolence 9 9 11 20 17 Confusion 1 6 2 0 4 Anxiety 3 0 2 0 3 Abnormal Gait 0 1 4 0 2 Dry Mouth 1 1 2 0 2 Nervousness 1 1 0 0 2 Vasodilatation 2 0 2 0 2 Hallucinations 0 1 2 2 1 Insomnia 0 1 2 0 1 Thinking Abnormal 0 1 2 0 1 Vertigo 1 0 0 0 1 Respiratory Dyspnea 2 3 6 5 4 Skin Pruritus 1 0 0 5 2 Rash 1 1 0 2 2 Sweating 1 1 2 2 2 Special Senses Abnormal Vision 1 0 2 0 2 The following adverse events not reflected in Table 3 occurred during titration with an overall frequency of 1% or greater and are listed in descending order of frequency within each body system.
Body as a Whole: Pain, fever, abdominal pain, chills, back pain, chest pain, infection
Cardiovascular: Migraine
Digestive: Diarrhea, dyspepsia, flatulence
Metabolic and Nutritional: Peripheral edema, dehydration
Nervous: Hypesthesia
Respiratory: Pharyngitis, cough increased
The following events occurred during titration with an overall frequency of less than 1% and are listed in descending order of frequency within each body system.
Body as a Whole: Flu syndrome, abscess, bone pain
Cardiovascular: Deep thrombophlebitis, hypertension, hypotension
Digestive: Anorexia, eructation, esophageal stenosis, fecal impaction, gum hemorrhage, mouth ulceration, oral moniliasis
Hemic and Lymphatic: Anemia, leukopenia
Metabolic and Nutritional: Edema, hypercalcemia, weight loss
Musculoskeletal: Myalgia, pathological fracture, myasthenia
Nervous: Abnormal dreams, urinary retention, agitation, amnesia, emotional lability, euphoria, incoordination, libido decreased, neuropathy, paresthesia, speech disorder
Respiratory: Hemoptysis, pleural effusion, rhinitis, asthma, hiccup, pneumonia, respiratory insufficiency, sputum increased
Skin and Appendages: Alopecia, exfoliative dermatitis
Special Senses: Taste perversion
Urogenital: Vaginal hemorrhage, dysuria, hematuria, urinary incontinence, urinary tract infection
A long-term extension study was conducted in 156 patients with malignancy and breakthrough cancer pain who were treated for an average of 129 days. Data are available for 152 of these patients. Table 4 lists by dose groups, adverse events with an overall frequency of 1% or greater that occurred during the long-term extension study and are commonly associated with opioid administration or are of particular clinical interest. Adverse events are listed in descending order of frequency within each body system.
Table 4.
Percent of Patients with Adverse Events Commonly Associated with
Opioid Administration or of Particular Clinical Interest Which Occurred
During Long-Term Treatment (Events in 1% or More of Patients)Dose Group 200-600 mcg 800-1400 mcg 1600 mcg >1600 mcg Any Number of Patients 98 83 53 27 152 Body As A Whole Asthenia 25 30 17 15 38 Headache 12 17 13 4 20 Accidental Injury 4 6 4 7 9 Hypertonia 2 2 2 0 3 Digestive Nausea 31 36 25 26 45 Vomiting 21 28 15 7 31 Constipation 14 11 13 4 20 Intestinal Obstruction 0 2 4 0 3 Cardiovascular Hypertension 1 1 0 0 1 Nervous Dizziness 12 10 9 0 16 Anxiety 9 8 8 7 15 Somnolence 8 13 8 7 15 Confusion 2 5 13 7 10 Depression 9 4 2 7 9 Insomnia 5 1 8 4 7 Abnormal Gait 5 1 0 0 4 Dry Mouth 3 1 2 4 4 Nervousness 2 2 0 4 3 Stupor 4 1 0 0 3 Vasodilatation 1 1 4 0 3 Thinking Abnormal 2 1 0 0 2 Abnormal Dreams 1 1 0 0 1 Convulsion 0 1 2 0 1 Myoclonus 0 0 4 0 1 Tremor 0 1 2 0 1 Vertigo 0 0 4 0 1 Respiratory Dyspnea 15 16 8 7 22 Skin Rash 3 5 8 4 8 Sweating 3 2 2 0 4 Pruritus 2 0 2 0 2 Special Senses Abnormal Vision 2 2 0 0 3 Urogenital Urinary Retention 1 2 0 0 2
The following events not reflected in Table 4 occurred with an overall frequency of 1% or greater in the long-term extension study and are listed in descending order of frequency within each body system.
Body as a Whole: Pain, fever, back pain, abdominal pain, chest pain, flu syndrome, chills, infection, abdomen enlarged, bone pain, ascites, sepsis, neck pain, viral infection, fungal infection, cachexia, cellulitis, malaise, pelvic pain
Cardiovascular: Deep thrombophlebitis, migraine, palpitation, vascular disorder
Digestive: Diarrhea, anorexia, dyspepsia, dysphagia, oral moniliasis, mouth ulceration, rectal disorder, stomatitis, flatulence, gastrointestinal hemorrhage, gingivitis, jaundice, periodontal abscess, eructation, glossitis, rectal hemorrhage
Hemic and Lymphatic: Anemia, leukopenia, thrombocytopenia, ecchymosis, lymphadenopathy, lymphedema, pancytopenia
Metabolic and Nutritional: Peripheral edema, edema, dehydration, weight loss, hyperglycemia, hypokalemia, hypercalcemia, hypomagnesemia
Musculoskeletal: Myalgia, pathological fracture, joint disorder, leg cramps, arthralgia, bone disorder
Nervous: Hypesthesia, paresthesia, hypokinesia, neuropathy, speech disorder
Respiratory: Cough increased, pharyngitis, pneumonia, rhinitis, sinusitis, bronchitis, epistaxis, asthma, hemoptysis, sputum increased
Skin and Appendages: Skin ulcer, alopecia
Special Senses: Tinnitus, conjunctivitis, ear disorder, taste perversion
Urogenital: Urinary tract infection, urinary incontinence, breast pain, dysuria, hematuria, scrotal edema, hydronephrosis, kidney failure, urinary urgency, urination impaired, breast neoplasm, vaginal hemorrhage, vaginitis
The following events occurred with a frequency of less than 1% in the long-term extension study and are listed in descending order of frequency within each body system.
Body as a Whole: Allergic reaction, cyst, face edema, flank pain, granuloma, bacterial infection, injection site pain, mucous membrane disorder, neck rigidity
Cardiovascular: Angina pectoris, hemorrhage, hypotension, peripheral vascular disorder, postural hypotension, tachycardia
Digestive: Cheilitis, esophagitis, fecal incontinence, gastroenteritis, gastrointestinal disorder, gum hemorrhage, hemorrhage of colon, hepatorenal syndrome, liver tenderness, tooth caries, tooth disorder
Hemic and Lymphatic: Bleeding time increased
Metabolic and Nutritional: Acidosis, generalized edema, hypocalcemia, hypoglycemia, hyponatremia, hypoproteinemia, thirst
Musculoskeletal: Arthritis, muscle atrophy, myopathy, synovitis, tendon disorder
Nervous: Acute brain syndrome, agitation, cerebral ischemia, facial paralysis, foot drop, hallucinations, hemiplegia, miosis, subdural hematoma
Respiratory: Hiccup, hyperventilation, lung disorder, pneumothorax, respiratory failure, voice alteration
Skin and Appendages: Herpes zoster, maculopapular rash, skin discoloration, urticaria, vesiculobullous rash
Special Senses: Ear pain, eye hemorrhage, lacrimation disorder, partial permanent deafness, partial transitory deafness
Urogenital: Kidney pain, nocturia, oliguria, polyuria, pyelonephritis
Post-Marketing Experience
The following adverse reactions have been identified during postapproval use of Actiq . Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Decisions to include these reactions in labeling are typically based on one or more of the following factors: (1) seriousness of the reaction, (2) frequency of the reporting, or (3) strength of causal connection to Actiq .
Digestive: Dental decay of varying severity including dental caries, tooth loss, and gum line erosion
DRUG ABUSE AND DEPENDENCE
Fentanyl is a mu-opioid agonist and a Schedule II controlled substance that can produce drug dependence of the morphine type. Actiq may be subject to misuse, abuse and addiction.
The administration of Actiq should be guided by the response of the patient. Physical dependence, per se, is not ordinarily a concern when one is treating a patient with chronic cancer pain, and fear of tolerance and physical dependence should not deter using doses that adequately relieve the pain.
Opioid analgesics may cause physical dependence. Physical dependence results in withdrawal symptoms in patients who abruptly discontinue the drug. Withdrawal also may be precipitated through the administration of drugs with opioid antagonist activity, e.g., naloxone, nalmefene, or mixed agonist/antagonist analgesics (pentazocine, butorphanol, buprenorphine, nalbuphine).
Physical dependence usually does not occur to a clinically significant degree until after several weeks of continued opioid usage. Tolerance, in which increasingly larger doses are required in order to produce the same degree of analgesia, is initially manifested by a shortened duration of analgesic effect, and subsequently, by decreases in the intensity of analgesia.
The handling of Actiq should be managed to minimize the risk of diversion, including restriction of access and accounting procedures as appropriate to the clinical setting and as required by law (see SAFETY AND HANDLING ).
OVERDOSAGE
Clinical Presentation
The manifestations of Actiq overdosage are expected to be similar in nature to intravenous fentanyl and other opioids, and are an extension of its pharmacological actions with the most serious significant effect being hypoventilation (see CLINICAL PHARMACOLOGY ).
General
Immediate management of opioid overdose includes removal of the Actiq unit, if still in the mouth, ensuring a patent airway, physical and verbal stimulation of the patient, and assessment of level of consciousness, ventilatory and circulatory status.
Treatment of Overdosage (Accidental Ingestion) in the Opioid NON-Tolerant Person
Ventilatory support should be provided, intravenous access obtained, and naloxone or other opioid antagonists should be employed as clinically indicated. The duration of respiratory depression following overdose may be longer than the effects of the opioid antagonist's action (e.g., the half-life of naloxone ranges from 30 to 81 minutes) and repeated administration may be necessary. Consult the package insert of the individual opioid antagonist for details about such use.
Treatment of Overdose in Opioid-Tolerant Patients
Ventilatory support should be provided and intravenous access obtained as clinically indicated. Judicious use of naloxone or another opioid antagonist may be warranted in some instances, but it is associated with the risk of precipitating an acute withdrawal syndrome.
General Considerations for Overdose
Management of severe Actiq overdose includes: securing a patent airway, assisting or controlling ventilation, establishing intravenous access, and GI decontamination by lavage and/or activated charcoal, once the patient's airway is secure. In the presence of hypoventilation or apnea, ventilation should be assisted or controlled and oxygen administered as indicated.
Patients with overdose should be carefully observed and appropriately managed until their clinical condition is well controlled.
Although muscle rigidity interfering with respiration has not been seen following the use of Actiq, this is possible with fentanyl and other opioids. If it occurs, it should be managed by the use of assisted or controlled ventilation, by an opioid antagonist, and as a final alternative, by a neuromuscular blocking agent.
DOSAGE AND ADMINISTRATION
Actiq is contraindicated in non-opioid tolerant individuals.
Actiq should be individually titrated to a dose that provides adequate analgesia and minimizes side effects (see Dose Titration ).
As with all opioids, the safety of patients using such products is dependent on health care professionals prescribing them in strict conformity with their approved labeling with respect to patient selection, dosing, and proper conditions for use.
Physicians and dispensing pharmacists must specifically question patients and caregivers about the presence of children in the home on a full time or visiting basis and counsel accordingly regarding the dangers to children of inadvertent exposure to Actiq.
Administration of Actiq
The blister package should be opened with scissors immediately prior to product use. The patient should place the Actiq unit in his or her mouth between the cheek and lower gum, occasionally moving the drug matrix from one side to the other using the handle. The Actiq unit should be sucked, not chewed. A unit dose of Actiq , if chewed and swallowed, might result in lower peak concentrations and lower bioavailability than when consumed as directed.
The Actiq unit should be consumed over a 15-minute period. Longer or shorter consumption times may produce less efficacy than reported in Actiq clinical trials. If signs of excessive opioid effects appear before the unit is consumed, the drug matrix should be removed from the patient's mouth immediately and future doses should be decreased.
Patients and caregivers must be instructed that Actiq contains medicine in an amount that could be fatal to a child. While all units should be disposed of immediately after use, partially used units represent a special risk and must be disposed of as soon as they are consumed and/or no longer needed. Patients and caregivers should be advised to dispose of any units remaining from a prescription as soon as they are no longer needed (see Disposal Instructions ).
Dose Titration
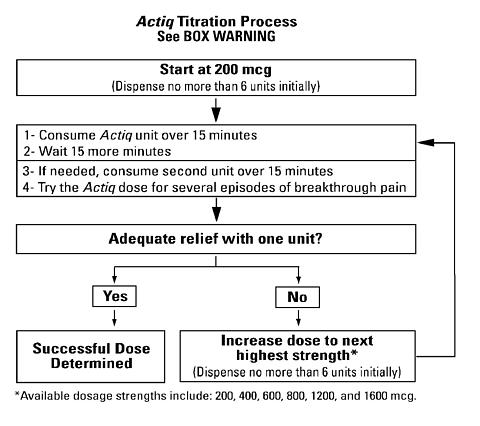
Starting Dose: The initial dose of Actiq to treat episodes of breakthrough cancer pain should be 200 mcg. Patients should be prescribed an initial titration supply of six 200 mcg Actiq units, thus limiting the number of units in the home during titration. Patients should use up all units before increasing to a higher dose.
From this initial dose, patients should be closely followed and the dosage level changed until the patient reaches a dose that provides adequate analgesia using a single Actiq dosage unit per breakthrough cancer pain episode.
Patients should record their use of Actiq over several episodes of breakthrough cancer pain and review their experience with their physicians to determine if a dosage adjustment is warranted.
Redosing Within a Single Episode: Until the appropriate dose is reached, patients may find it necessary to use an additional Actiq unit during a single episode. Redosing may start 15 minutes after the previous unit has been completed (30 minutes after the start of the previous unit). While patients are in the titration phase and consuming units which individually may be subtherapeutic, no more than two units should be taken for each individual breakthrough cancer pain episode.
Increasing the Dose: If treatment of several consecutive breakthrough cancer pain episodes requires more than one Actiq per episode, an increase in dose to the next higher available strength should be considered. At each new dose of Actiq during titration, it is recommended that six units of the titration dose be prescribed. Each new dose of Actiq used in the titration period should be evaluated over several episodes of breakthrough cancer pain (generally 1-2 days) to determine whether it provides adequate efficacy with acceptable side effects. The incidence of side effects is likely to be greater during this initial titration period compared to later, after the effective dose is determined.
Daily Limit: Once a successful dose has been found (i.e., an average episode is treated with a single unit), patients should limit consumption to four or fewer units per day. If consumption increases above four units/day, the dose of the long-acting opioid used for persistent cancer pain should be re-evaluated.
Dosage Adjustment
Experience in a long-term study of Actiq used in the treatment of breakthrough cancer pain suggests that dosage adjustment of both Actiq and the maintenance (around-the-clock) opioid analgesic may be required in some patients to continue to provide adequate relief of breakthrough cancer pain.
Generally, the Actiq dose should be increased when patients require more than one dosage unit per breakthrough cancer pain episode for several consecutive episodes. When titrating to an appropriate dose, small quantities (six units) should be prescribed at each titration step. Physicians should consider increasing the around-the-clock opioid dose used for persistent cancer pain in patients experiencing more than four breakthrough cancer pain episodes daily.
Discontinuation of Actiq
For patients requiring discontinuation of opioids, a gradual downward titration is recommended because it is not known at what dose level the opioid may be discontinued without producing the signs and symptoms of abrupt withdrawal.
SAFETY AND HANDLING
Actiq is supplied in individually sealed child-resistant blister packages. The amount of fentanyl contained in Actiq can be fatal to a child. Patients and their caregivers must be instructed to keep Actiq out of the reach of children (see BOX WARNING, WARNINGS, PRECAUTIONS, and PATIENT LEAFLET ).
Store at 20°-25°C (68°-77°F) with excursions permitted between 15° and 30°C (59° to 86°F) until ready to use. (See USP Controlled Room Temperature.)
Actiq should be protected from freezing and moisture. Do not use if the blister package has been opened.
DISPOSAL OF ACTIQ
Patients must be advised to dispose of any units remaining from a prescription as soon as they are no longer needed. While all units should be disposed of immediately after use, partially consumed units represent a special risk because they are no longer protected by the child-resistant blister package, yet may contain enough medicine to be fatal to a child (see Information for Patients ).
A temporary storage bottle is provided as part of the Actiq Welcome Kit (see Information for Patients and Their Caregivers ). This container is to be used by patients or their caregivers in the event that a partially consumed unit cannot be disposed of promptly. Instructions for usage of this container are included in the patient leaflet.
Patients and members of their household must be advised to dispose of any units remaining from a prescription as soon as they are no longer needed. Instructions are included in Information for Patients and Their Caregivers and in the patient leaflet. If additional assistance is required, referral to the Actiq 800# (1-800-896-5855) should be made.
HOW SUPPLIED
Actiq is supplied in six dosage strengths. Each unit is individually wrapped in a child-resistant, protective blister package. These blister packages are packed 30 per shelf carton for use when patients have been titrated to the appropriate dose.
Patients should be prescribed an initial titration supply of six 200 mcg Actiq units. At each new dose of Actiq during titration, it is recommended that only six units of the next higher dose be prescribed.
Each dosage unit has a white to off-white color. The dosage strength of each unit is marked on the solid drug matrix, the handle tag, the blister package and the carton. See blister package and carton for product information.
Dosage Strength
(fentanyl base)Carton/Blister
Package ColorNDC Number 200 mcg Gray NDC 63459-502-30 400 mcg Blue NDC 63459-504-30 600 mcg Orange NDC 63459-506-30 800 mcg Purple NDC 63459-508-30 1200 mcg Green NDC 63459-512-30 1600 mcg Burgundy NDC 63459-516-30 Note: Colors are a secondary aid in product identification. Please be sure to confirm the printed dosage before dispensing.
Rx only.
DEA order form required. A Schedule C-II narcotic.
Manufactured by:
Cephalon, Inc., Salt Lake City, UT 84116, USA
U. S. Patent No. 4,671,953, 4,863,737 and 5,785,989
Printed in USA
#1598.01
©2000, 2001, 2003, 2004 Cephalon, Inc. All rights reserved.
Subscribe to the "News" RSS Feed 
TOP ۞
