-
Humalog-Pen (Lilly)
DESCRIPTION
Humalog® (insulin lispro, rDNA origin) is a human insulin analog that is a rapid-acting, parenteral blood glucose-lowering agent. Chemically, it is Lys(B28), Pro(B29) human insulin analog, created when the amino acids at positions 28 and 29 on the insulin B-chain are reversed. Humalog is synthesized in a special non-pathogenic laboratory strain of Escherichia coli bacteria that has been genetically altered by the addition of the gene for insulin lispro.
Humalog has the following primary structure:
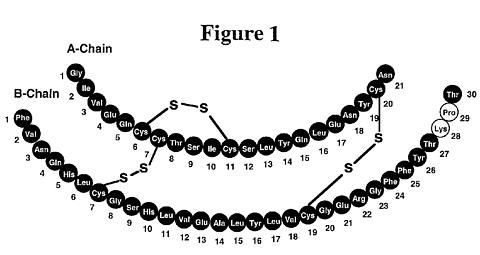
Insulin lispro has the empirical formula C 257 H 383 N 65 O 77 S 6 and a molecular weight of 5808, both identical to that of human insulin.
The vials, cartridges, and Pens contain a sterile solution of Humalog for use as an injection. Humalog injection consists of zinc-insulin lispro crystals dissolved in a clear aqueous fluid.
Each milliliter of Humalog injection contains insulin lispro 100 Units, 16 mg glycerin, 1.88 mg dibasic sodium phosphate, 3.15 mg Metacresol, zinc oxide content adjusted to provide 0.0197 mg zinc ion, trace amounts of phenol, and water for injection. Insulin lispro has a pH of 7.0 to 7.8. Hydrochloric acid 10% and/or sodium hydroxide 10% may be added to adjust pH.
CLINICAL PHARMACOLOGY
Antidiabetic Activity
The primary activity of insulin, including Humalog, is the regulation of glucose metabolism. In addition, all insulins have several anabolic and anti-catabolic actions on many tissues in the body. In muscle and other tissues (except the brain), insulin causes rapid transport of glucose and amino acids intracellularly, promotes anabolism, and inhibits protein catabolism. In the liver, insulin promotes the uptake and storage of glucose in the form of glycogen, inhibits gluconeogenesis, and promotes the conversion of excess glucose into fat.
Humalog has been shown to be equipotent to human insulin on a molar basis. One unit of Humalog has the same glucose-lowering effect as one unit of human regular insulin, but its effect is more rapid and of shorter duration. The glucose-lowering activity of Humalog and human regular insulin is comparable when administered to normal volunteers by the intravenous route.
Pharmacokinetics
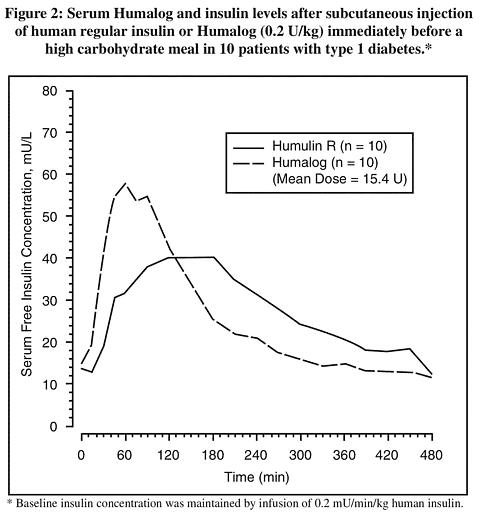
Absorption and Bioavailability -- Humalog is as bioavailable as human regular insulin, with absolute bioavailability ranging between 55% to 77% with doses between 0.1 to 0.2 U/kg, inclusive. Studies in normal volunteers and patients with type 1 (insulin-dependent) diabetes demonstrated that Humalog is absorbed faster than human regular insulin (U-100) ( see Figure 2). In normal volunteers given subcutaneous doses of Humalog ranging from 0.1 to 0.4 U/kg, peak serum levels were seen 30 to 90 minutes after dosing. When normal volunteers received equivalent doses of human regular insulin, peak insulin levels occurred between 50 to 120 minutes after dosing. Similar results were seen in patients with type 1 diabetes. The pharmacokinetic profiles of Humalog and human regular insulin are comparable to one another when administered to normal volunteers by the intravenous route. Humalog was absorbed at a consistently faster rate than human regular insulin in healthy male volunteers given 0.2 U/kg human regular insulin or Humalog at abdominal, deltoid, or femoral subcutaneous sites, the three sites often used by patients with diabetes. After abdominal administration of Humalog, serum drug levels are higher and the duration of action is slightly shorter than after deltoid or thigh administration ( see DOSAGE AND ADMINISTRATION ). Humalog has less intra- and inter-patient variability compared to human regular insulin.
Distribution -- The volume of distribution for Humalog is identical to that of human regular insulin, with a range of 0.26 to 0.36 L/kg.
Metabolism -- Human metabolism studies have not been conducted. However, animal studies indicate that the metabolism of Humalog is identical to that of human regular insulin.
Elimination -- When Humalog is given subcutaneously, its t 1/2 is shorter than that of human regular insulin (1 vs. 1.5 hours, respectively). When given intravenously, Humalog and human regular insulin show identical dose-dependent elimination, with a t 1/2 of 26 and 52 minutes at 0.1 U/kg and 0.2 U/kg, respectively.
Pharmacodynamics
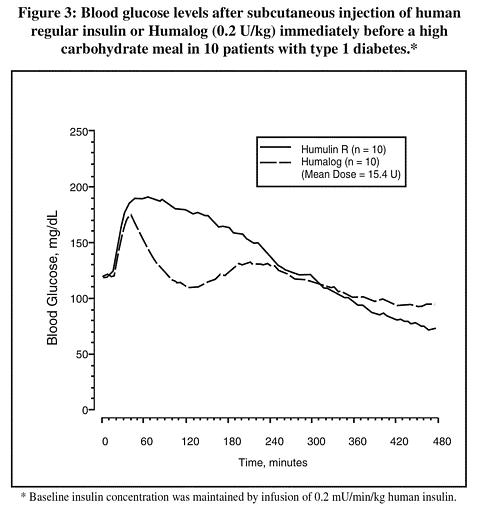
Studies in normal volunteers and patients with diabetes demonstrated that Humalog has a more rapid onset of glucose-lowering activity, an earlier peak for glucose-lowering, and a shorter duration of glucose-lowering activity than human regular insulin ( see Figure 3). The earlier onset of activity of Humalog is directly related to its more rapid rate of absorption. The time course of action of insulin and insulin analogs, such as Humalog, may vary considerably in different individuals or within the same individual. The parameters of Humalog activity (time of onset, peak time, and duration) as designated in Figure 3 should be considered only as general guidelines. The rate of insulin absorption and consequently the onset of activity is known to be affected by the site of injection, exercise, and other variables ( see PRECAUTIONS , General ).
Special Populations
Age and Gender -- Information on the effect of age and gender on the pharmacokinetics of Humalog is unavailable. However, in large clinical trials, subgroup analysis based on age and gender did not indicate any difference in postprandial glucose parameters between Humalog and human regular insulin.
Smoking -- The effect of smoking on the pharmacokinetics and pharmacodynamics of Humalog has not been studied.
Pregnancy -- The effect of pregnancy on the pharmacokinetics and pharmacodynamics of Humalog has not been studied.
Obesity -- The effect of obesity and/or subcutaneous fat thickness on the pharmacokinetics and pharmacodynamics of Humalog has not been studied. In large clinical trials, which included patients with Body Mass Index up to and including 35 kg/m 2 , no consistent differences were seen between Humalog and Humulin® R with respect to postprandial glucose parameters.
Renal Impairment -- Some studies with human insulin have shown increased circulating levels of insulin in patients with renal failure. In a study of 25 patients with type 2 diabetes and a wide range of renal function, the pharmacokinetic differences between Humalog and human regular insulin were generally maintained. However, the sensitivity of the patients to insulin did change, with an increased response to insulin as the renal function declined. Careful glucose monitoring and dose adjustments of insulin, including Humalog, may be necessary in patients with renal dysfunction.
Hepatic Impairment -- Some studies with human insulin have shown increased circulating levels of insulin in patients with hepatic failure. In a study of 22 patients with type 2 diabetes, impaired hepatic function did not affect the subcutaneous absorption or general disposition of Humalog when compared to patients with no history of hepatic dysfunction. In that study, Humalog maintained its more rapid absorption and elimination when compared to human regular insulin. Careful glucose monitoring and dose adjustments of insulin, including Humalog, may be necessary in patients with hepatic dysfunction.
Clinical Studies
In open-label, cross-over studies of 1008 patients with type 1 diabetes and 722 patients with type 2 (non-insulin-dependent) diabetes, Humalog reduced postprandial glucose compared with human regular insulin ( see Table 1). The clinical significance of improvement in postprandial hyperglycemia has not been established.
Table 1 Comparison of Means of Glycemic Parameters at the End of Combined Treatment Periods. All Randomized Patients in Cross-Over Studies (3 months for each treatment)Type 1, N=1008
Glycemic Parameter,
(mg/dL)
Humalog a
Humulin R a *Fasting Blood Glucose209.5 ± 91.6 204.1 ± 89.3 1-Hour Postprandial232.4 ± 97.7 250.0 ± 96.7 2-Hour Postprandial200.9 ± 95.4 231.7 ± 103.9 HbA 1c (%)8.2 ± 1.5 8.2 ± 1.5 Type 2, N=722
Glycemic Parameter,
(mg/dL)Humalog a Humulin R a Fasting Blood Glucose192.1 ± 67.9 183.1 ± 66.1 1-Hour Postprandial238.1 ± 79.7 250.0 ± 75.2 2-Hour Postprandial217.4 ± 83.2 236.5 ± 80.6 HbA 1c (%)8.2 ± 1.3 8.2 ± 1.4 a Mean ± Standard Deviation.*Humulin R (human insulin [rDNA origin] injection).
In 12-month parallel studies in patients with type 1 and type 2 diabetes, HbA 1c did not differ between patients treated with human regular insulin and those treated with Humalog.
Hypoglycemia -- While the overall rate of hypoglycemia did not differ between patients with type 1 and type 2 diabetes treated with Humalog compared with human regular insulin, patients with type 1 diabetes treated with Humalog had fewer hypoglycemic episodes between midnight and 6 a.m. The lower rate of hypoglycemia in the Humalog-treated group may have been related to higher nocturnal blood glucose levels, as reflected by a small increase in mean fasting blood glucose levels.
Humalog in Combination with Sulfonylurea Agents -- In a two-month study in patients with fasting hyperglycemia despite maximal dosing with sulfonylureas (SU), patients were randomized to one of three treatment regimens; Humulin® NPH at bedtime plus SU, Humalog three times a day before meals plus SU, or Humalog three times a day before meals and Humulin NPH at bedtime. The combination of Humalog and SU resulted in an improvement in HbA 1c accompanied by a weight gain ( see Table 2).
Table 2 Results of a Two-Month Study in Which Humalog
Was Added to Sulfonylurea Therapy in Patients Not
Adequately Controlled on Sulfonylurea AloneHumulin N
h.s.+ SUHumalog a.c.+SU Humalog
a.c. + Humulin N h.s.Randomized (n)135 139 149 HbA lc (%) at
baseline9.9 10.0 10.0 HbA lc (%) at
2-months8.7 8.4 8.5 HbA lc (%) change
from baseline-1.2 -1.6 -1.4 Weight gain at
2-months (kg)0.6 1.2 1.5 Hypoglycemia *
(events/mo)0.11 0.03 0.09 Number of injections1 3 4 Total insulin dose
(U/kg) at 2-months0.23 0.33 0.52 a.c.-three times a day before meals, h.s.-at bedtime, SU-oral sulfonylurea agent.*blood glucose </= 36 mg/dL or needing assistance from third party.
Humalog in External Insulin Pumps -- To evaluate the administration of Humalog via external insulin pumps, two open-label cross-over design studies were performed in patients with type 1 diabetes. One study involved 39 patients treated for 24 weeks with Humalog or regular human insulin. After 12 weeks of treatment, the mean HbA 1c values decreased from 7.8% to 7.2% in the Humalog-treated patients and from 7.8% to 7.5% in the regular insulin-treated patients. Another study involved 60 patients treated for 24 weeks with either Humalog or buffered regular human insulin. After 12 weeks of treatment, the mean HbA 1c values decreased from 7.7% to 7.4% in the Humalog-treated patients and remained unchanged from 7.7% in the buffered regular insulin-treated patients. Rates of hypoglycemia were comparable between treatment groups in both studies. Humalog administration in insulin pumps has not been studied in patients with type 2 diabetes.
INDICATIONS AND USAGE
Humalog is an insulin analog that is indicated in the treatment of patients with diabetes mellitus for the control of hyperglycemia. Humalog has a more rapid onset and a shorter duration of action than human regular insulin. Therefore, in patients with type 1 diabetes, Humalog should be used in regimens that include a longer-acting insulin. However, in patients with type 2 diabetes, Humalog may be used without a longer-acting insulin when used in combination therapy with sulfonylurea agents.
Humalog may be used in an external insulin pump, but should not be diluted or mixed with any other insulin when used in the pump.
CONTRAINDICATIONS
Humalog is contraindicated during episodes of hypoglycemia and in patients sensitive to Humalog or one of its excipients.
WARNINGS
This human insulin analog differs from human regular insulin by its rapid onset of action as well as a shorter duration of activity. When used as a mealtime insulin, the dose of Humalog should be given within 15 minutes before or immediately after the meal. Because of the short duration of action of Humalog, patients with type 1 diabetes also require a longer-acting insulin to maintain glucose control (except when using an external insulin pump). Glucose monitoring is recommended for all patients with diabetes and is particularly important for patients using an external insulin pump.
Hypoglycemia is the most common adverse effect associated with insulins, including Humalog. As with all insulins, the timing of hypoglycemia may differ among various insulin formulations. Glucose monitoring is recommended for all patients with diabetes.
Any change of insulin should be made cautiously and only under medical supervision. Changes in insulin strength, manufacturer, type (e.g., regular, NPH, analog), species (animal, human), or method of manufacture (rDNA vs. animal-source insulin) may result in the need for a change in dosage.
External Insulin Pumps: When used in an external insulin pump, Humalog should not be diluted or mixed with any other insulin. Patients should carefully read and follow the external insulin pump manufacturer's instructions and the "INFORMATION FOR THE PATIENT" insert before using Humalog.
Physicians should carefully evaluate information on external insulin pump use in this Humalog physician package insert and in the external insulin pump manufacturer's instructions. If unexplained hyperglycemia or ketosis occurs during external insulin pump use, prompt identification and correction of the cause is necessary. The patient may require interim therapy with subcutaneous insulin injections ( see PRECAUTIONS , For Patients Using External Insulin Pumps , and DOSAGE AND ADMINISTRATION ).
PRECAUTIONS
General
Hypoglycemia and hypokalemia are among the potential clinical adverse effects associated with the use of all insulins. Because of differences in the action of Humalog and other insulins, care should be taken in patients in whom such potential side effects might be clinically relevant (e.g., patients who are fasting, have autonomic neuropathy, or are using potassium-lowering drugs or patients taking drugs sensitive to serum potassium level). Lipodystrophy and hypersensitivity are among other potential clinical adverse effects associated with the use of all insulins.
As with all insulin preparations, the time course of Humalog action may vary in different individuals or at different times in the same individual and is dependent on site of injection, blood supply, temperature, and physical activity.
Adjustment of dosage of any insulin may be necessary if patients change their physical activity or their usual meal plan. Insulin requirements may be altered during illness, emotional disturbances, or other stresses.
Hypoglycemia -- As with all insulin preparations, hypoglycemic reactions may be associated with the administration of Humalog. Rapid changes in serum glucose levels may induce symptoms of hypoglycemia in persons with diabetes, regardless of the glucose value. Early warning symptoms of hypoglycemia may be different or less pronounced under certain conditions, such as long duration of diabetes, diabetic nerve disease, use of medications such as beta-blockers, or intensified diabetes control.
Renal Impairment -- The requirements for insulin may be reduced in patients with renal impairment.
Hepatic Impairment -- Although impaired hepatic function does not affect the absorption or disposition of Humalog, careful glucose monitoring and dose adjustments of insulin, including Humalog, may be necessary.
Allergy -- Local Allergy -- As with any insulin therapy, patients may experience redness, swelling, or itching at the site of injection. These minor reactions usually resolve in a few days to a few weeks. In some instances, these reactions may be related to factors other than insulin, such as irritants in a skin cleansing agent or poor injection technique.
Systemic Allergy -- Less common, but potentially more serious, is generalized allergy to insulin, which may cause rash (including pruritus) over the whole body, shortness of breath, wheezing, reduction in blood pressure, rapid pulse, or sweating. Severe cases of generalized allergy, including anaphylactic reaction, may be life threatening. In controlled clinical trials, pruritus (with or without rash) was seen in 17 patients receiving Humulin R (N=2969) and 30 patients receiving Humalog (N=2944) (p=0.053). Localized reactions and generalized myalgias have been reported with the use of cresol as an injectable excipient.
Antibody Production -- In large clinical trials, antibodies that cross-react with human insulin and insulin lispro were observed in both Humulin R- and Humalog-treatment groups. As expected, the largest increase in the antibody levels during the 12-month clinical trials was observed with patients new to insulin therapy.
Usage in External Insulin Pumps -- The infusion set (reservoir syringe, tubing, and catheter), Disetronic® D-TRON® 2 , 3 or D-TRON® 2 , 3 plus cartridge adapter, and Humalog in the external insulin pump reservoir should be replaced and a new infusion site selected every 48 hours or less. Humalog in the external insulin pump should not be exposed to temperatures above 37°C (98.6°F).
In the D-TRON® 2 , 3 or D-TRON® 2 , 3 plus pump, Humalog 3 mL cartridges may be used for up to 7 days. However, as with other external insulin pumps, the infusion set should be replaced and a new infusion site should be selected every 48 hours or less.
When used in an external insulin pump, Humalog should not be diluted or mixed with any other insulin ( see INDICATIONS AND USAGE , WARNINGS , PRECAUTIONS , For Patients Using External Insulin Pumps , Mixing of Insulins , DOSAGE AND ADMINISTRATION , and Storage ).
Information for Patients
Patients should be informed of the potential risks and advantages of Humalog and alternative therapies. Patients should also be informed about the importance of proper insulin storage, injection technique, timing of dosage, adherence to meal planning, regular physical activity, regular blood glucose monitoring, periodic glycosylated hemoglobin testing, recognition and management of hypo- and hyperglycemia, and periodic assessment for diabetes complications.
Patients should be advised to inform their physician if they are pregnant or intend to become pregnant.
Refer patients to the "INFORMATION FOR THE PATIENT" insert for information on proper injection technique, timing of Humalog dosing (</=15 minutes before or immediately after a meal), storing and mixing insulin, and common adverse effects.
Use of the Humalog Pen: Patients should read the "INFORMATION FOR THE PATIENT" insert and the "Disposable Insulin Delivery Device User Manual" before starting therapy with a Humalog Pen and re-read them each time the prescription is renewed. Patients should be instructed on how to properly use the delivery device (refer to "Disposable Insulin Delivery Device User Manual"), prime the Pen, and properly dispose of needles. Patients should be advised not to share their Pens with others.
For Patients Using External Insulin Pumps: Patients using an external infusion pump should be trained in intensive insulin therapy and in the function of their external insulin pump and pump accessories. Humalog may be used with the MiniMed® 1 Models 506, 507, and 508 insulin pumps using MiniMed® 1 Polyfin® 1 infusion sets. Humalog may also be used in Disetronic® 2 H-TRONplus® V100 insulin pump (with plastic 3.15 mL insulin reservoir), and the Disetronic D-TRON® 2 , 3 and D-TRON® 2 , 3 plus insulin pumps (with Humalog 3 mL cartridges) using Disetronic Rapid® 2 infusion sets.
The infusion set (reservoir syringe, tubing, catheter), D-TRON® 2 , 3 or D-TRON® 2 , 3 plus cartridge adapter, and Humalog in the external insulin pump reservoir should be replaced, and a new infusion site selected every 48 hours or less. Humalog in the external pump should not be exposed to temperatures above 37°C (98.6°F). A Humalog 3 mL cartridge used in the D-TRON® 2 , 3 or D-TRON® 2 , 3 plus pump should be discarded after 7 days, even if it still contains Humalog. Infusion sites that are erythematous, pruritic, or thickened should be reported to medical personnel, and a new site selected.
Humalog should not be diluted or mixed with any other insulin when used in an external insulin pump.
Laboratory Tests
As with all insulins, the therapeutic response to Humalog should be monitored by periodic blood glucose tests. Periodic measurement of glycosylated hemoglobin is recommended for the monitoring of long-term glycemic control.
Drug Interactions
Insulin requirements may be increased by medications with hyperglycemic activity such as corticosteroids, isoniazid, certain lipid-lowering drugs (e.g., niacin), estrogens, oral contraceptives, phenothiazines, and thyroid replacement therapy ( see CLINICAL PHARMACOLOGY ).
Insulin requirements may be decreased in the presence of drugs with hypoglycemic activity, such as oral hypoglycemic agents, salicylates, sulfa antibiotics, and certain antidepressants (monoamine oxidase inhibitors), certain angiotensin-converting-enzyme inhibitors, beta-adrenergic blockers, inhibitors of pancreatic function (e.g., octreotide), and alcohol. Beta-adrenergic blockers may mask the symptoms of hypoglycemia in some patients.
Mixing of Insulins -- Care should be taken when mixing all insulins as a change in peak action may occur. The American Diabetes Association warns in its Position Statement on Insulin Administration, "On mixing, physiochemical changes in the mixture may occur (either immediately or over time). As a result, the physiological response to the insulin mixture may differ from that of the injection of the insulins separately." Mixing Humalog with Humulin N or Humulin® U does not decrease the absorption rate or the total bioavailability of Humalog. Given alone or mixed with Humulin N, Humalog results in a more rapid absorption and glucose-lowering effect compared with human regular insulin.
The effects of mixing Humalog with insulins of animal source or insulin preparations produced by other manufacturers have not been studied ( see WARNINGS ).
If Humalog is mixed with a longer-acting insulin, such as Humulin N or Humulin U, Humalog should be drawn into the syringe first to prevent clouding of the Humalog by the longer-acting insulin. Injection should be made immediately after mixing. Mixtures should not be administered intravenously.
Humalog should not be diluted or mixed with any other insulin when used in an external insulin pump.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals have not been performed to evaluate the carcinogenic potential of Humalog. Humalog was not mutagenic in a battery of in vitro and in vivo genetic toxicity assays (bacterial mutation tests, unscheduled DNA synthesis, mouse lymphoma assay, chromosomal aberration tests, and a micronucleus test). There is no evidence from animal studies of Humalog-induced impairment of fertility.
Pregnancy
Teratogenic Effects -- Pregnancy Category B -- Reproduction studies have been performed in pregnant rats and rabbits at parenteral doses up to 4 and 0.3 times, respectively, the average human dose (40 units/day) based on body surface area. The results have revealed no evidence of impaired fertility or harm to the fetus due to Humalog. There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Although there are limited clinical studies of the use of Humalog in pregnancy, published studies with human insulins suggest that optimizing overall glycemic control, including postprandial control, before conception and during pregnancy improves fetal outcome. Although the fetal complications of maternal hyperglycemia have been well documented, fetal toxicity also has been reported with maternal hypoglycemia. Insulin requirements usually fall during the first trimester and increase during the second and third trimesters. Careful monitoring of the patient is required throughout pregnancy. During the perinatal period, careful monitoring of infants born to mothers with diabetes is warranted.
Nursing Mothers
It is unknown whether Humalog is excreted in significant amounts in human milk. Many drugs, including human insulin, are excreted in human milk. For this reason, caution should be exercised when Humalog is administered to a nursing woman. Patients with diabetes who are lactating may require adjustments in Humalog dose, meal plan, or both.
Pediatric Use
In a 9-month, cross-over study of pre-pubescent children (n=60), aged 3 to 11 years, comparable glycemic control as measured by HbA 1c was achieved regardless of treatment group: human regular insulin 30 minutes before meals 8.4%, Humalog immediately before meals 8.4%, and Humalog immediately after meals 8.5%. In an 8-month, cross-over study of adolescents (n=463), aged 9 to 19 years, comparable glycemic control as measured by HbA 1c was achieved regardless of treatment group; human regular insulin 30 to 45 minutes before meals 8.7% and Humalog immediately before meals 8.7%. The incidence of hypoglycemia was similar for all three treatment regimens. Adjustment of basal insulin may be required. To improve accuracy in dosing in pediatric patients, a diluent may be used. If the diluent is added directly to the Humalog vial, the shelf-life may be reduced ( see DOSAGE AND ADMINISTRATION ).
Geriatric Use
Of the total number of subjects (n=2834) in eight clinical studies of Humalog, twelve percent (n=338) were 65 years of age or over. The majority of these were type 2 patients. HbA 1c values and hypoglycemia rates did not differ by age. Pharmacokinetic / Pharmacodynamic studies to assess the effect of age on the onset of Humalog action have not been performed.
ADVERSE REACTIONS
Clinical studies comparing Humalog with human regular insulin did not demonstrate a difference in frequency of adverse events between the two treatments.
Adverse events commonly associated with human insulin therapy include the following:
Body as a Whole -- allergic reactions ( see PRECAUTIONS ).
Skin and Appendages -- injection site reaction, lipodystrophy, pruritus, rash.
Other -- hypoglycemia ( see WARNINGS and PRECAUTIONS ).
OVERDOSAGE
Hypoglycemia may occur as a result of an excess of insulin relative to food intake, energy expenditure, or both. Mild episodes of hypoglycemia usually can be treated with oral glucose. Adjustments in drug dosage, meal patterns, or exercise, may be needed. More severe episodes with coma, seizure, or neurologic impairment may be treated with intramuscular/subcutaneous glucagon or concentrated intravenous glucose. Sustained carbohydrate intake and observation may be necessary because hypoglycemia may recur after apparent clinical recovery.
DOSAGE AND ADMINISTRATION
Humalog is intended for subcutaneous administration, including use in select external insulin pumps ( see DOSAGE AND ADMINISTRATION , External Insulin Pumps ). Dosage regimens of Humalog will vary among patients and should be determined by the Health Care Professional familiar with the patient's metabolic needs, eating habits, and other lifestyle variables. Pharmacokinetic and pharmacodynamic studies showed Humalog to be equipotent to human regular insulin (i.e., one unit of Humalog has the same glucose-lowering capability as one unit of human regular insulin), but with more rapid activity. The quicker glucose-lowering effect of Humalog is related to the more rapid absorption rate from subcutaneous tissue. An adjustment of dose or schedule of basal insulin may be needed when a patient changes from other insulins to Humalog, particularly to prevent pre-meal hyperglycemia.
When used as a meal-time insulin, Humalog should be given within 15 minutes before or immediately after a meal. Human regular insulin is best given 30 to 60 minutes before a meal. To achieve optimal glucose control, the amount of longer-acting insulin being given may need to be adjusted when using Humalog.
The rate of insulin absorption and consequently the onset of activity is known to be affected by the site of injection, exercise, and other variables. Humalog was absorbed at a consistently faster rate than human regular insulin in healthy male volunteers given 0.2 U/kg human regular insulin or Humalog at abdominal, deltoid, or femoral sites, the three sites often used by patients with diabetes. When not mixed in the same syringe with other insulins, Humalog maintains its rapid onset of action and has less variability in its onset of action among injection sites compared with human regular insulin ( see PRECAUTIONS ). After abdominal administration, Humalog concentrations are higher than those following deltoid or thigh injections. Also, the duration of action of Humalog is slightly shorter following abdominal injection, compared with deltoid and femoral injections. As with all insulin preparations, the time course of action of Humalog may vary considerably in different individuals or within the same individual. Patients must be educated to use proper injection techniques.
Humalog may be diluted with STERILE DILUENT for Humalog® , Humulin® N, Humulin® 50/50, Humulin® 70/30, and NPH Iletin® to a concentration of 1:10 (equivalent to U-10) or 1:2 (equivalent to U-50). Diluted Humalog may remain in patient use for 28 days when stored at 5°C (41°F) and for 14 days when stored at 30°C (86°F). Do not dilute Humalog when used in an external insulin pump.
Parenteral drug products should be inspected visually prior to administration whenever the solution and the container permit. If the solution is cloudy, contains particulate matter, is thickened, or is discolored, the contents must not be injected. Humalog should not be used after its expiration date.
External Insulin Pumps -- Humalog may be used with MiniMed® 1 Models 506, 507, and 508 insulin pumps using MiniMed® 1 Polyfin® 1 infusion sets. Humalog may also be used in the Disetronic® 2 H-TRONplus® V100 insulin pump (with plastic 3.15 mL insulin reservoir) and the Disetronic D-TRON® 2 , 3 and D-TRON® 2 , 3 plus pumps (with Humalog 3 mL cartridges) using Disetronic Rapid® 2 infusion sets.
Humalog should not be diluted or mixed with any other insulin when used in an external insulin pump.
HOW SUPPLIED
Humalog (insulin lispro injection, rDNA origin) vials are available in the following package size:
100 units per mL (U-100)
10 mL vials NDC 0002-7510-01 (VL-7510)
Humalog (insulin lispro injection, rDNA origin) cartridges are available in the following package size:
5 X 3 mL cartridges 3 NDC 0002-7516-59 (VL-7516)
Humalog (insulin lispro injection, rDNA origin) Pen, disposable insulin delivery device, is available in the following package size:
5 X 3 mL disposable
insulin delivery devices NDC 0002-8725-59 (HP-8725)
1 MiniMed® and Polyfin® are registered trademarks of MiniMed, Inc.
2 Disetronic®, H-TRONplus®, D-TRON®, and Rapid® are registered trademarks of Roche Diagnostics GMBH.
3 3 mL cartridge is for use in Owen Mumford, Ltd.'s Autopen® 3 mL insulin delivery device and Disetronic D-TRON® and D-TRON®plus pumps. Autopen® is a registered trademark of Owen Mumford, Ltd. Other product and company names may be the trademarks of their respective owners.
Storage -- Unopened Humalog should be stored in a refrigerator (2° to 8°C [36° to 46°F]), but not in the freezer. Do not use Humalog if it has been frozen. Unrefrigerated (below 30°C [86°F]) vials, cartridges, and Pens must be used within 28 days or be discarded, even if they still contain Humalog. Protect from direct heat and light. See table below:
Not in-use (unopened) Room Temperature below 86°F (30°C) Not in-use (unopened) Refrigerated In-use (opened) Room Temperature, below
86°F (30°C)10 mL Vial28 days Until expiration date 28 days, refrigerated/room temperature. 3 mL Cartridge28 days Until expiration date 28 days, Do not refrigerate. 3 mL Pen28 days Until expiration date 28 days, Do not refrigerate.
Use in an External Insulin Pump -- A Humalog 3 mL cartridge used in the D-TRON® 2 , 3 or D-TRON® 2 , 3 plus should be discarded after 7 days, even if it still contains Humalog. Infusion sets, D-TRON® 2 , 3 and D-TRON® 2 , 3 plus cartridge adapters, and Humalog in the external insulin pump reservoir should be discarded every 48 hours or less.
Literature revised August 4, 2004
Manufactured by Lilly France S.A.S.
F-67640 Fegersheim, France
for Eli Lilly and Company
Indianapolis, IN 46285, USA
Copyright © 1996, 2004, Eli Lilly and Company. All rights reserved.
PA 9129 FSAMP
Subscribe to the "News" RSS Feed 
TOP ۞
