-
Ultralytic 2 Foam 20% (Ivax Dermatologicals)
DESCRIPTION
Ultralytic™ 2 Foam contains 20% urea in a topical cosmetic vehicle containing 12% ammonium lactate.
Ultralytic™ 2 Foam is a keratolytic emollient moisturizer. Each gram contains 20% urea, purified water, ammonium lactate, white petrolatum, octyl palmitate, caprylic/capric trigylceride, hydrogenated polyisobutene, propylene glycol, rice starch, polysorbate 60, cyclomethycone, glyceryl stearate & PEG-100 stearate, cetearyl alcohol & cetearyl glucoside, polysorbate -20, phenoxyethanol, cetyl alcohol, dimethicone, potassium sorbate, allantoin, tocopheryl acetate, xanthan gum. In propellants isobutane & propane & butane.
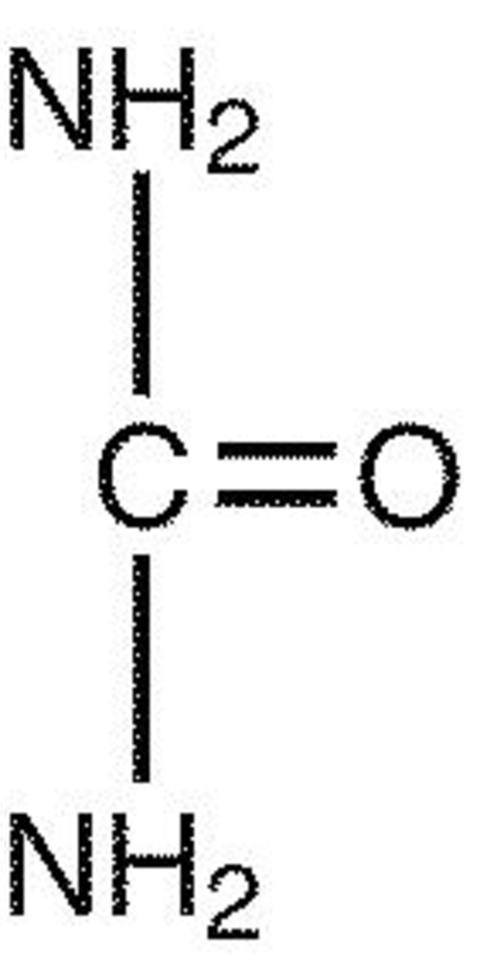
Urea is a diamide of carbonic acid with the following chemical structure:
CLINICAL PHARMACOLOGY
Urea gently lyses/dissolves the intercellular matrix of surface skin cells loosening and allowing a shedding of rough, thickened and scaly hyperkeratotic skin. Urea also moisturizes and softens skin.
PHARMACOKINETICS:
The mechanisms of action of topically applied Urea are not yet known.
INDICATIONS AND USES
For softening, smoothing and removing rough scaling hyperkeratotic skin in conditions such as xerosis, ichthyosis, skin cracks and fissures, dermatitis, eczema, psoriasis, keratoses and calluses.
CONTRAINDICATIONS
Known hypersensitivity to any of the listed ingredients. Discontinue if hypersensitivity is observed. Sun exposure to areas of skin treated with Ultralytic™ 2 Foam should be minimized or avoided.
WARNINGS
For external use only. Avoid contact with eyes, lips or mucous membranes.
PRECAUTIONS
Information for Patients: This medication is to be used as directed by a physician and should not be used to treat any condition other than that for which it was prescribed. If redness or irritation occurs, discontinue use.
PREGNANCY: Pregnancy Category C. Animal reproduction studies have not been conducted with Ultralytic™ 2 Foam . It is also not known whether Ultralytic™ 2 Foam can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Ultralytic™ 2 Foam should be given to a pregnant woman only if clearly needed.
NURSING MOTHERS: It is not known whether or not this drug is secreted in human milk. Because many drugs are secreted in human milk, caution should be exercised when Ultralytic™ 2 Foam is administered to a nursing woman.
ADVERSE REACTIONS
Transient stinging, burning, itching or irritation may occur and normally disappear on discontinuing the medication.
DOSAGE AND ADMINISTRATION
Apply Ultralytic™ 2 Foam to affected skin twice per day, or as directed by a physician. Rub in until completely absorbed.
HOW SUPPLIED
Ultralytic™ 2 Foam 20% is supplied in a 100 g (3.5 oz.) canister.
NDC 13613-730-35
Store at room temperature 15° - 25°C (59° - 77° F).
Protect from freezing.
Mfg. Exclusively for:
Ivax Dermatologicals, Inc.
4400 Biscayne Blvd.
Miami, FL 33137
Mfg. by:
Agis Industries, (1983) Ltd.
Yeruham, 80500 Israel
05/05
Subscribe to the "News" RSS Feed 
TOP ۞
